La NASA quiere más mujeres en la ciencia
Por Agencia EFE
–
22/11/2011
Washington,
22 nov (EFE).- La NASA quiere más mujeres en la ciencia y con el fin de
animar a las jóvenes estudiantes a que encaminen sus carreras hacia las
ramas de ingeniería, matemáticas y tecnología ha creado una nueva web
que fue presentada hoy.
La página women.nasa.gov/a2i/
contiene vídeos en los que pueden saber más sobre las mujeres que
trabajan en la NASA, sus carreras, su formación y cómo comenzaron a
trabajar para la agencia espacial estadounidense.
Como la
matemática Carolina Restrepo, quien creció en Colombia y Bolivia, y a
los 18 años no dudó en marcharse a Estados Unidos para estudiar
ingeniería, con la ilusión de poder diseñar, construir y hacer volar un
aeroplano radiocontrolado.
Fue lo que vio en la televisión sobre
la NASA lo que le impulsó a querer trabajar en la agencia espacial.
"Nunca había tenido la oportunidad de conocer o hablar con alguien que
había trabajado en el campo del espacio, pero no me importó, me
concentré para que cuando creciera sucediera", cuenta en su relato.
Pocas
semanas después de comenzar su primer semestre en la Universidad de
Texas A&M, leyó que la NASA estaba contratando gente de su edad y
ese se convirtió en su nuevo objetivo. Un año más tarde estaba
trabajando en el Centro Espacial Johnson de Houston algo que siempre ha
disfrutado desde entonces.
Como estudiante trabajó en varios
proyectos, pero cuenta que sus favoritos eran los que estaban
relacionados con diseñar algoritmos sobre el vuelo de las naves
espaciales, un trabajo que requirió una base sólida en Matemáticas y en
Física para poder simular el vuelo del vehículo espacial.
Ahora
forma parte del equipo de diseño de la cápsula Orion, el nuevo vehículo
de la NASA con el que espera poder enviar al hombre a Marte.
"Trabajar
en la NASA hace que todos esos años de trabajo duro y las largas noches
de estudio hayan merecido la pena. No cambiaría esto por nada en el
mundo", añadió.
Con esta herramienta "tenemos la oportunidad de
llegar a la próxima generación e inspirar a las jóvenes de hoy a
perseguir una carrera en la ciencia y la tecnología", señaló Rebecca
Keiser, directora adjunta de políticas de integración y representante de
la NASA ante la Casa Blanca del consejo de políticas para mujeres y
niñas.
La web incluye cuatro cuentas de Twitter, una por cada uno
de los temas, en las que las estudiantes pueden interactuar enviando sus
preguntas a las científicas.
La NASA ya contaba con una página web women.nasa.gov,
en la que se incluyen el testimonio de mujeres en puestos destacados en
la agencia, entre otras, la subdirectora Lori Garver, con el mismo
objetivo de llevar la ciencia a la nueva generación.
Según datos
de la NASA, de los 18.544 empleados que tiene la agencia espacial en
todos sus centros, 6.539 son mujeres, de las que 417 son hispanas.
©
EFE 2011. Está expresamente prohibida la redistribución y la redifusión
de todo o parte de los contenidos de los servicios de Efe, sin previo y
expreso consentimiento de la Agencia EFE S.A.
EFE
Antarctic Ozone Hole Slightly Smaller than Average This
Year
The Antarctic ozone hole reached its maximum
single-day area for 2013 on Sept. 16. The ozone hole (purple and blue) is the
region over Antarctica with total ozone at or below 220 Dobson units (a common
unit for measuring ozone concentration).
Image Credit: NASA's Goddard Space Flight
Center
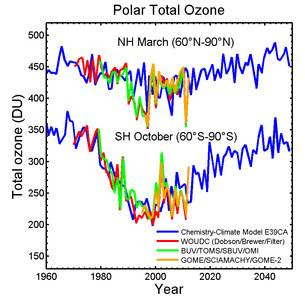
The ozone hole that forms each year in the stratosphere over Antarctica was
slightly smaller in 2013 than average in recent decades, according to NASA
satellite data.
The ozone hole is a seasonal phenomenon that starts to form during the
Antarctic spring (August and September). The September-October 2013 average size
of the hole was 8.1 million square miles (21 million square kilometers). For
comparison, the average size measured since the mid-1990s when the annual
maximum size stopped growing is 8.7 million square miles (22.5 million square
kilometers). However, the size of the hole in any particular year is not enough
information for scientists to determine whether a healing of the hole has
begun.
"There was a lot of Antarctic ozone depletion in 2013, but because of above
average temperatures in the Antarctic lower stratosphere, the ozone hole was a
bit below average compared to ozone holes observed since 1990," said Paul
Newman, an atmospheric scientist and ozone expert at NASA's Goddard Space Flight
Center in Greenbelt, Md.
The ozone hole forms when the sun begins rising again after several months of
winter darkness. Polar-circling winds keep cold air trapped above the continent,
and sunlight-sparked reactions involving ice clouds and chlorine from manmade
chemicals begin eating away at the ozone. Most years, the conditions for ozone
depletion ease before early December when the seasonal hole closes.
Levels of most ozone-depleting chemicals in the atmosphere have gradually
declined as the result of the 1987 Montreal Protocol, an international treaty to
protect the ozone layer by phasing out production of ozone-depleting chemicals.
As a result, the size of the hole has stabilized, with variation from year to
year driven by changing meteorological conditions.
Daily images from Jul. 1 to Oct. 15 show the
evolution of the 2013 ozone hole. The ozone hole maximum occurred on Sept. 16,
2013.
Image Credit: NASA/Robert Simmon/Ozone Hole
Watch
Image Token:
The single-day maximum area this year was reached on Sept. 16 when the
maximum area reached 9.3 million square miles (24 million square kilometers),
about equal to the size of North America. The largest single-day ozone hole
since the mid-1990s was 11.5 million square miles (29.9 million square
kilometers) on Sept. 9, 2000.
Science teams from NASA and the National Oceanic and Atmospheric
Administration (NOAA) have been monitoring the ozone layer from the ground and
with a variety of instruments on satellites and balloons since the 1970s. These
ozone instruments capture different aspects of ozone depletion. The independent
analyses ensure that the international community understands the trends in this
critical part of Earth's atmosphere. The resulting views of the ozone hole have
differences in the computation of the size of the ozone hole, its depth, and
record dates.
NASA observations of the ozone hole during 2013 were produced from data
supplied by the Ozone Monitoring Instrument on NASA's Aura satellite and the
Ozone Monitoring and Profiler Suite instrument on the NASA-NOAA Suomi National
Polar-orbiting Partnership satellite. Long-term satellite ozone-monitoring
instruments have included the Total Ozone Mapping Spectrometer, the second
generation Solar Backscatter Ultraviolet Instrument, the Stratospheric Aerosol
and Gas Experiment series of instruments, and the Microwave Limb Sounder.
Related Links:
NASA
Guillermo Gonzalo Sánchez Achutegui